Introduction
The HCN Lewis structure is an essential concept in basic chemistry that helps explain how atoms in hydrogen cyanide (HCN) bond together and share electrons. Understanding this structure is crucial for students who are learning about chemical bonding, molecular geometry, and electron distribution. In this detailed article, we will walk through every aspect of the HCN molecule’s Lewis structure, break down step-by-step how to draw it, explain its shape and polarity, and explore why this information matters in chemistry.
This comprehensive guide will help you fully grasp the fundamentals and nuances of the HCN Lewis structure in a simple and clear way.
What Is the Lewis Structure?
The Lewis structure, also known as a Lewis dot diagram, is a visual representation showing how the valence electrons are arranged around atoms in a molecule. These diagrams help us visualize bonding pairs (shared electrons between atoms) and lone pairs (electrons not involved in bonding), giving insight into the stability and behavior of molecules.
When we look at the HCN molecule, the Lewis structure reveals how atoms share electrons to reach a more stable state.
Understanding the HCN Molecule
Before drawing the Lewis structure, it’s important to know what atoms make up hydrogen cyanide and what their roles are.
Hydrogen cyanide (HCN) consists of:
- One hydrogen atom (H)
- One carbon atom (C)
- One nitrogen atom (N)
The chemical formula for hydrogen cyanide is HCN. Although it is a simple molecule, its bonding pattern is unique and instructive.
Hydrogen cyanide is a colorless liquid or gas at room temperature and is known for its strong toxicity. From a chemical perspective, it is used in many industrial processes and organic synthesis reactions.
Step-by-Step Guide to Drawing the HCN Lewis Structure
Drawing a Lewis structure requires systematic steps. Let’s go through each stage carefully.
Step 1: Count Total Valence Electrons
Valence electrons are the electrons in the outermost shell of an atom. These electrons are responsible for bonding.
The atoms in HCN have the following valence electrons:
- Hydrogen (H) has 1 valence electron
- Carbon (C) has 4 valence electrons
- Nitrogen (N) has 5 valence electrons
Total valence electrons = 1 (H) + 4 (C) + 5 (N) = 10 valence electrons
These 10 electrons must be arranged in a way that satisfies the octet rule as much as possible.
Step 2: Draw a Skeleton Structure
In most molecules, the least electronegative atom (except hydrogen) becomes the central atom. In HCN, carbon is less electronegative than nitrogen and can form more bonds, so carbon becomes the central atom.
The skeleton structure is:
H — C — N
At this point, we connect the atoms with single bonds:
- A single bond between H and C
- A single bond between C and N
Each single bond uses 2 electrons. Since there are two bonds, that consumes 4 electrons.
Remaining electrons = 10 total – 4 used = 6 electrons left
Step 3: Assign Remaining Electrons as Lone Pairs
We place the remaining 6 electrons around the more electronegative atom first, which is nitrogen. This gives nitrogen 3 lone pairs.
The structure now looks like:
H — C — N:
(with 3 lone pairs on nitrogen)
At this stage, hydrogen has a full shell with 2 electrons, nitrogen has 8 electrons (2 from the bond and 6 from lone pairs), but carbon only has 4 (from two single bonds). Carbon needs 8 electrons to satisfy the octet.
Step 4: Form Multiple Bonds to Complete Octet
To satisfy the octet rule for carbon, we must form additional bonds. We can increase bonding between carbon and nitrogen by converting lone pairs on nitrogen into bonding pairs.
By forming a triple bond between carbon and nitrogen, we effectively share more electrons:
H — C ≡ N
Here, carbon shares 6 electrons with nitrogen, which gives carbon a total of 8 electrons (2 from hydrogen bond and 6 from nitrogen bond), fulfilling the octet.
Nitrogen also achieves an octet and retains one lone pair.
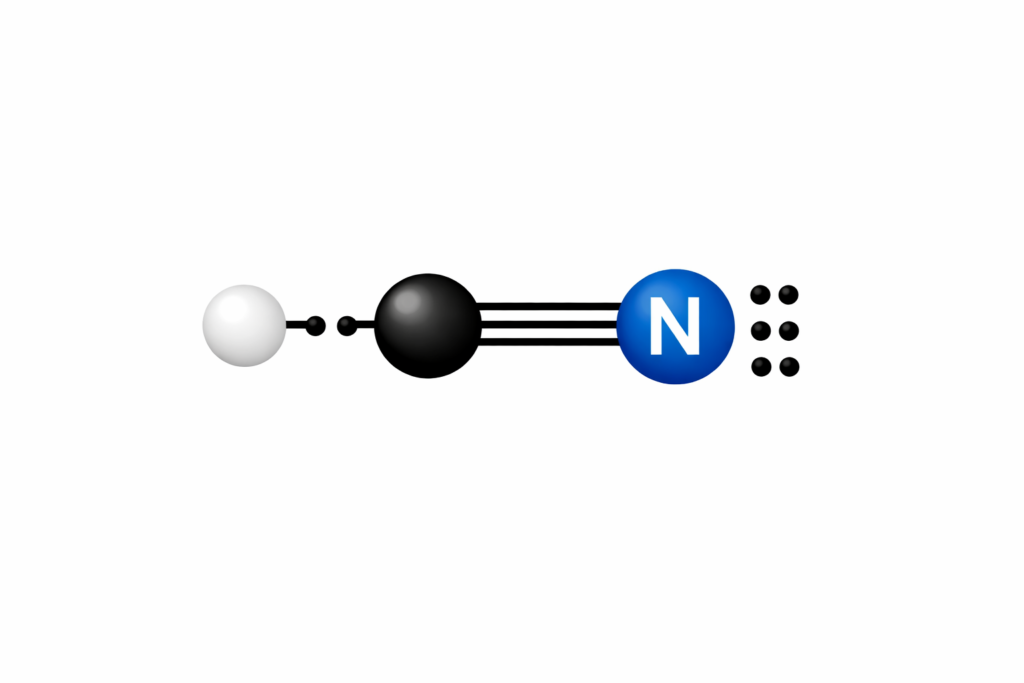
Completed Lewis Structure
The final Lewis structure is:
H — C ≡ N:
- Hydrogen: 2 electrons (full outer shell)
- Carbon: 8 electrons (full octet)
- Nitrogen: 8 electrons (full octet)
This structure uses all 10 valence electrons and satisfies the octet rule for carbon and nitrogen.
Formal Charge Calculation
Formal charge helps determine whether the drawn Lewis structure is stable and represents the real electron distribution in the molecule.
Formal charge is calculated as:
Formal Charge = (Valence electrons of atom) − (Nonbonding electrons + 1/2 bonding electrons)
Let’s check formal charges on each atom in HCN:
- Hydrogen: (1) – (0 + 1) = 0
- Carbon: (4) – (0 + 4) = 0
- Nitrogen: (5) – (2 + 3) = 0
Since all atoms have a formal charge of zero, the structure is stable and correct.
Molecular Geometry and Shape
The VSEPR (Valence Shell Electron Pair Repulsion) theory helps predict the shape of molecules based on electron pair repulsion.
In HCN:
- Carbon has two regions of electron density (one bond with hydrogen, one triple bond with nitrogen)
- There are no lone pairs on carbon
This gives the molecule a linear geometry, with a bond angle of approximately 180 degrees.
The linear structure explains the physical and chemical properties of HCN, including its polarity.
Polarity of HCN
A molecule is polar if it has an uneven distribution of charge or if the bond dipoles do not cancel out.
In HCN:
- Hydrogen has lower electronegativity
- Carbon has higher electronegativity than hydrogen
- Nitrogen has higher electronegativity than carbon
The electronegativity difference creates a dipole moment that points from hydrogen toward nitrogen.
Because the molecule is linear, the dipole movements do not cancel, making HCN a polar molecule. This polarity explains why HCN dissolves in some polar solvents and affects its reactivity.
Bond Order and Strength
The bond order refers to the number of bonds between atoms.
In HCN:
- C — H is a single bond, so bond order is 1
- C ≡ N is a triple bond, so bond order is 3
Higher bond order means stronger bonds. Therefore, the C≡N triple bond in HCN is very strong and requires more energy to break compared to a single bond.
This bond strength affects how HCN behaves in chemical reactions, making certain reactions slower unless specific conditions are met.
Why Understanding HCN Lewis Structure Matters
Learning how to draw the HCN Lewis structure builds foundational skills that apply to many areas in chemistry:
- Predicting reactivity
- Understanding polarity and solubility
- Predicting molecular shape and physical properties
- Analyzing bond strength and stability
These skills are useful not just for simple molecules like HCN but for more complex organic and inorganic compounds.
Common Mistakes Students Make
Many students struggle with drawing Lewis structures correctly. Here are common pitfalls:
- Not counting the total valence electrons accurately
- Placing hydrogen in the center of the structure
- Forgetting to satisfy the octet rule for carbon or nitrogen
- Ignoring formal charge calculations
Avoiding these mistakes improves confidence and accuracy in structural chemistry.
Conclusion
The HCN Lewis structure is more than just dots and lines. It reveals how atoms share electrons to form stable bonds, how shape and polarity arise, and how molecular properties are determined at a fundamental level.
By carefully counting valence electrons, assigning bonds and lone pairs, and checking for formal charges, you can confidently draw and understand Lewis structures for many molecules beyond HCN.
Understanding this topic lays the groundwork for future studies in chemical bonding, molecular geometry, spectroscopy, and reaction mechanisms.